|
Unfortunately for Meyer, Mendeleev’s table was published first in 1869 and Meyer’s not until 1870. Mendeleev and German chemist Lothar Meyer, working independently of one another, both produced remarkably similar results concerning the periodic nature of the elements. Many scientists contributed, and there is even some disagreement concerning who deserves credit for being the “father” of the periodic table. Mendeleev is generally credited with the production of the first periodic table, but it was hardly a solo effort. In 1913, the English chemist Henry Moseley found that by arranging the elements in the periodic table by atomic number rather than atomic mass, the problems with the order of the elements was resolved. Mendeleev’s periodic table was not completely accurate, and new discoveries indicated that several of the elements in his table were not in the correct order. Mendeleev’s predictions were proven to be correct with the discoveries of scandium, gallium, and germanium. Undaunted, he predicted the existence, as well as the properties, of the unknown elements that would be found to fill these spaces. However, his procedure left several empty spaces in his periodic table. When organized this way he noticed the periodic pattern in their properties and formulated the periodic law. Using the standards from the conference, the Russian chemist, Dimitri Mendeleev, created a table of the 60 elements known at the time by arranging them by increasing atomic mass. Development of the Periodic TableĪs the result of the Karlsruhe Conference held in Germany in 1860, the atomic masses used by chemists throughout the world were standardized. In addition, Newlands was criticized for his analogy of elements to musical notes by fellow scientists who thought it to be unscientific. 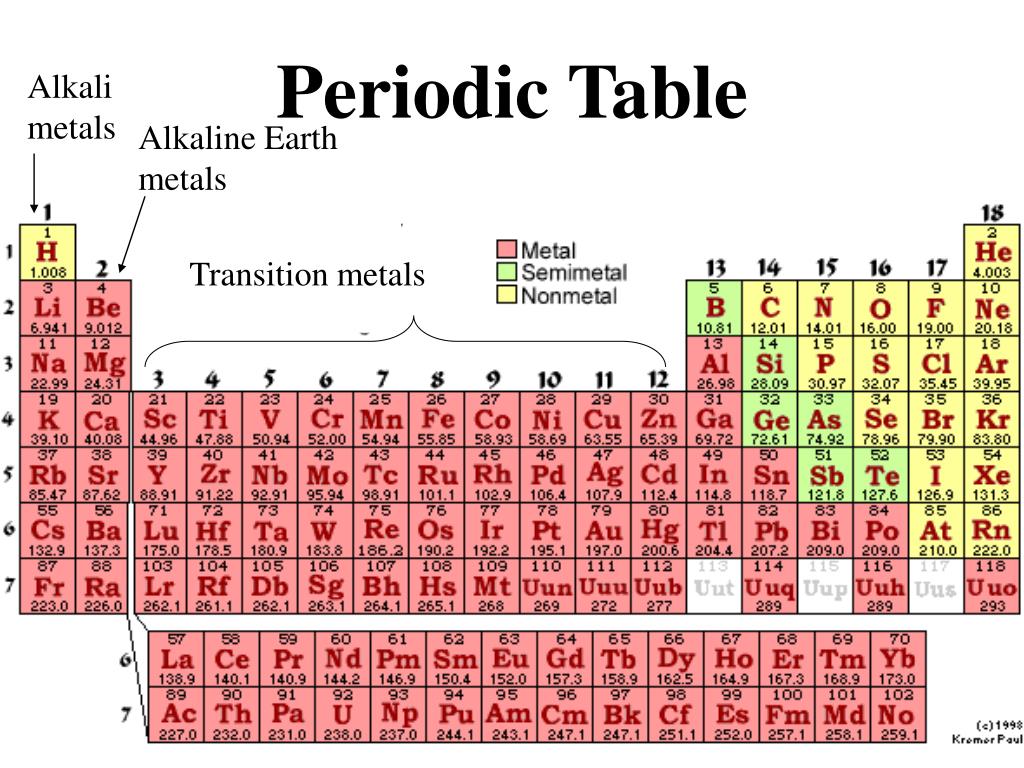
The law of octaves was not readily accepted because it did not work for all of the known elements. Because an octave is a group of musical notes that repeats every eighth note, Newlands named his discovery the law of octaves. Beginning with hydrogen, he observed that every eighth element had similar properties. The grouping of elements into triads appeared to Dobereiner to be more than a mere coincidence, and he developed the law of triads from 1817 to 1829.įrom 1863 to 1865, English chemist John Newlands found that when the elements were arranged according to increasing atomic mass, there was a repeating pattern relating to the chemical properties of the elements. In each group, the atomic weight of the middle element was the approximate average of the atomic weights of the other two elements. One example was calcium, strontium, and barium another group was chlorine, bromine, and iodine. He observed that certain elements could be grouped together in threes based on their similarities. One of the earliest attempts to organize the elements based on their chemical and physical properties was made by the German chemist, Johann Dobereiner. What was needed was a standardized method for determining atomic mass, and a system of classification of the elements grouping them by similarities.Ī number of chemists began to observe relationships among the properties of certain elements. 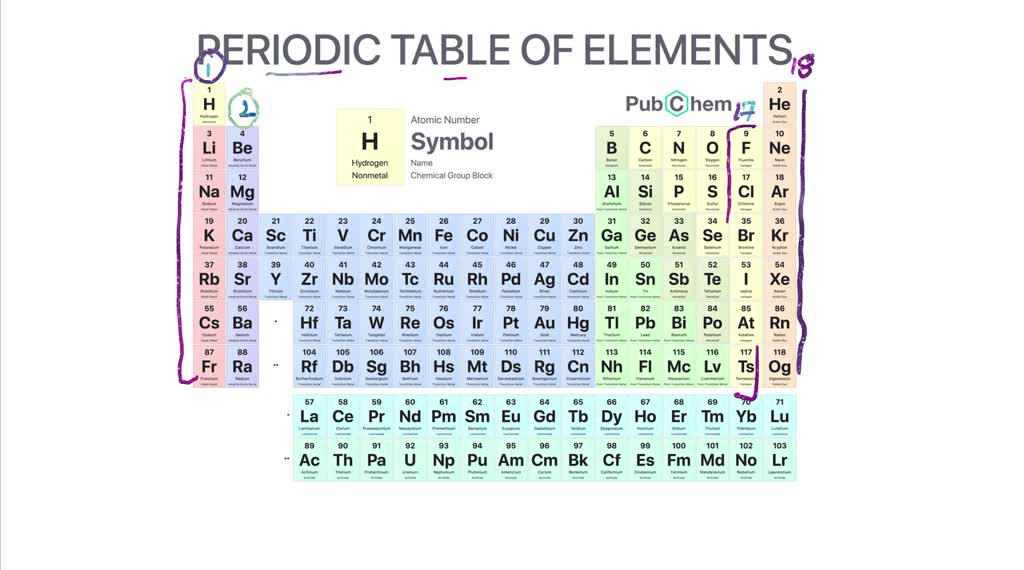
There was no method for accurately determining atomic masses, and chemists used different mass numbers in their work, making the results of one chemist’s work difficult, if not impossible, to be interpreted by another. The structure of the nucleus including protons and neutronsīy the early-1800s, approximately 60 elements had been discovered, and chemists were overwhelmed with the task of categorizing the properties of so many new elements.In this lesson we will discuss a brief history of the development of the periodic table, and how to use the modern periodic table to obtain information about the atoms of the known elements. ⬅ Previous Lesson Workshop Index Next Lesson ➡Ītomic Structure, Periodicity, and Matter:The Periodic Table Objective
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
